linsitinib
OSI 906
ASP7487
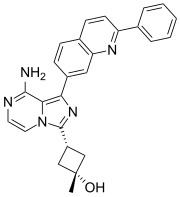
3-[8-Amino-1-(2-phenyl-7-quinolyl)imidazo[1,5-a]pyrazin-3-yl]-1-methyl-cyclobutanol
CAS: 867160-71-2
Chemical Formula: C26H23N5O
Molecular Weight: 421.5
Elemental Analysis: C, 74.09; H, 5.50; N, 16.62; O, 3.80
PHASE 2
Linsitinib (OSI-906) is an orally bioavailable small molecule inhibitor of the insulin-like growth factor 1 receptor (IGF-1R) with potential antineoplastic activity. OSI-906 selectively inhibits IGF-1R, which may result in the inhibition of tumor cell proliferation and the induction of tumor cell apoptosis. Overexpressed in a variety of human cancers, IGFR-1 stimulates cell proliferation, enables oncogenic transformation, and suppresses apoptosis
Linsitinib (OSI-906) was developed through drug-discovery efforts focused on identifying a potent and selective, small-molecule inhibitor of the IGF-1R signaling axis. The lead optimization phase utilized IR and IGF-1R co-crystal structures, with lead compounds from the imidazopyrazine series, to afford a structure-based design-driven component, which complemented ongoing empirical medicinal chemistry efforts. These combined approaches improved metabolic and pharmacokinetic liabilities of earlier lead compounds and ultimately led to the discovery of OSI-906. OSI-906 was synthesized from an advanced imidazopyrazine intermediate in two linear steps. OSI-906 potently inhibits ligand-dependent auto-phosphorylation of both human IGF-1R and IR in cells, while displaying a high degree of selectivity versus a wide panel of protein kinases.
Moreover, OSI-906, through its inhibition of both IGF-1R and IR, prevents ligand-induced activation of downstream pathways including pAKT, pERK1/2 and p-p70S6K and, therefore, inhibits proliferation in a variety of tumor cell lines. Robust anti-tumor activity was achieved in an IGF-1R-driven LISN xenograft model following once-daily oral administration of OSI-906. The anti-tumor activity obtained in this study correlated well with the degree and duration of inhibition of tumor IGF-1R phosphorylation achieved in vivo by OSI-906. OSI-906 is a novel, potent, selective and orally bioavailable dual IGF-1R/IR kinase inhibitor with demonstrated in vivo efficacy in tumor models. It is currently being evaluated in clinical trials.
Furthermore, the exceptional selectivity profile of OSI-906 in conjunction with its ability to inhibit both IGF-1R and IR provides the unique opportunity to fully target the IGF-1R/IR axis. (source: Future Medicinal Chemistry September 2009, Vol. 1, No. 6, Pages 1153-1171. )
Linsitinib is an experimental drug candidate for the treatment of various types of cancer. It is an inhibitor of the insulin receptor and of the insulin-like growth factor 1 receptor (IGF-1R).[1] This prevents tumor cell proliferation and induces tumor cell apoptosis.[2]
The development of target-based anti-cancer therapies has become the focus of a large number of pharmaceutical research and development programs. Various strategies of intervention include targeting protein tyrosine kinases, including receptor tyrosine kinases believed to drive or mediate tumor growth.
Insulin-like growth factor-1 receptor (IGF-1R) is a receptor tyrosine kinase that plays a key role in tumor cell proliferation and apoptosis inhibition, and has become an attractive cancer therapy target. IGF-1R is involved in the establishment and maintenance of cellular transformation, is frequently overexpressed by human tumors, and activation or overexpression thereof mediates aspects of the malignant phenotype. IGF-1R activation increases invasion and metastasis propensity.
Inhibition of receptor activation has been an attractive method having the potential to block IGF-mediated signal transduction. Anti-IGF-1R antibodies to block the extracellular ligand-binding portion of the receptor and small molecules to target the enzyme activity of the tyrosine kinase domain have been developed. See Expert Opin. Ther. Patents, 17(1):25-35 (2007); Expert Opin. Ther. Targets, 12(5):589-603 (2008); and Am J. Transl. Res., 1:101-114 (2009).
US 2006/0235031 (published Oct. 19, 2006) describes a class of bicyclic ring substituted protein kinase inhibitors, including Example 31 thereof, which corresponds to the dual IR/IGF-1R inhibitor known as OSI-906. As of 2011, OSI-906 is in clinical development in various cancers and tumor types. The preparation and characterization of OSI-906, which can be named as cis-3-[8-amino-1-(2-phenyl-quinolin-7-yl)-imidazo[1,5-a]pyrazin-3-yl]-1-methylcyclobutanol, is described in the aforementioned US 2006/0235031.
OSI-906 is a potent, selective, and orally bioavailable dual IGF-1R/IR kinase inhibitor with favorable drug-like properties. The selectivity profile of OSI-906 in conjunction with its ability to inhibit both IGF-1R and IR affords the special opportunity to fully target the IGF-1R/IR axis. See Future Med. Chem., 1(6), 1153-1171, (2009).
It is desirable to develop novel processes to prepare imidazopyrazine compounds, namely OSI-906, which may be practical, economical, efficient, reproducible, large scale, and meet regulatory requirements.
Linsitinib was discovered by OSI Pharmaceuticals and is currently in Phase III clinical trials for adrenocortical carcinoma and Phase II clinical trials for lung and ovarian cancers.[3][4]
- Mulvihill, MJ; Cooke, A; Rosenfeld-Franklin, M; Buck, E; Foreman, K; Landfair, D; O’Connor, M; Pirritt, C et al. (2009). “Discovery of OSI-906: A selective and orally efficacious dual inhibitor of the IGF-1 receptor and insulin receptor”. Future medicinal chemistry 1 (6): 1153–71. doi:10.4155/fmc.09.89.PMID 21425998.
- “Linsitinib”. NCI Drug Dictionary. National Cancer Institute. Retrieved October 16, 2012.
- “OSI Pharmaceuticals, LLC”. Astellas Pharma. Retrieved October 16, 2012.
- “Linsitinib”. National Institutes of Health’s clinicaltrials.gov. Retrieved October 16, 2012.
OSI-906: A novel, potent, and selective first-in-class small molecule insulin-like growth factor 1 receptor (IGF-1R) inhibitor in phase I clinical trials
238th ACS Natl Meet (August 16-20, Washington) 2009, Abst MEDI 152
238th ACS Natl Meet (August 16-20, Washington) 2009, Abst MEDI 152
………………………………………………………
EXAMPLES1
cis-3-[8-amino-1-(2-phenyl-quinolin-7-yl)-imidazo[1,5-a]pyrazin-3-yl]-1-methylcyclobutanol (OSI-906) (Compound 1)
A vessel was charged with DMF (79 kg), cis-3-(8-amino-1-bromo-imidazo[1,5-a]pyrazin-3-yl)-1-methylcyclobutanol (16.725 kg), 2-phenyl-7-(4,4,5,5-tetramethyl-[1,3,2]dioxaborolan-2-yl)-quinoline (22.4 kg), triphenylphosphine (0.586 kg), cesium carbonate (36.7 kg) and water (20.1 kg). The reaction mixture was degassed and heated to 95-105° C. and a solution of palladium acetate (0.125 kg) in DMF (9.8 kg) was added and rinsed in with DMF (5.9 kg). After the reaction was complete, water (154 kg) was added keeping the temperature above 70° C. The resultant slurry was cooled and the solid was collected by filtration. After washing with a mixture of DMF (9.4 kg) and water (23.4 kg) and then water (67 kg) the solid was suspended in water (167 kg) at 50° C. and the pH of the mixture was adjusted to 2.9 with 6N hydrochloric acid (10.9 kg). The resultant yellow slurry was filtered to remove the major impurities and the cake was washed with water (67 kg). The acid solution was stirred at 50-55° C. and polymer bound trimercaptotriazine resin (MP-TMT) (4.9 kg) was added. The mixture was stirred for 23 hours, the resin was removed by filtration and the cake was washed with water (58 kg).
The resultant acid solution was diluted with 2-propanol (82 kg), the temperature was adjusted to 35-45° C. and the pH was adjusted to 5.0 by the addition of 1N sodium hydroxide solution. The mixture was cooled, the yellow product was collected by filtration and was washed with water (33 kg). The solid was re-suspended in water (157 kg) stirred, filtered and washed with water (125 kg). The solid was dried under vacuum at 45-55° C. (the resulting material was a hemihydrate of OSI-906 designated Form C) and was then stirred in refluxing 2-propanol (157 kg) for 3 hours. The mixture was cooled and the solid was isolated by filtration. After washing with 2-propanol (26.7 kg), the product was dried at 45-55° C. under vacuum to yield 15.6 kg (65% yield) of OSI-906. The resulting material was an anhydrous crystalline form of OSI-906 designated Form A.
Example 2cis-3-(1-bromo-8-chloro-imidazo[1,5-a]pyrazin-3-yl)-1-methylcyclobutanol
THF (87 kg) and 3M methyl magnesium chloride (83.6 kg) were charged to a vessel. The contents were cooled to −65 to −55° C. and 3-(1-bromo-8-chloro-imidazo[1,5-a]pyrazin-3-yl)-cyclobutanone (33.0 kg) in THF (253 kg) was added, maintaining the temperature at −65° C. to −45° C.
The charged vessel was rinsed with THF (41 kg) and the reaction mixture was stirred at −65 to −45° C. until reaction completion. Preferably, the level of iron present in the reaction is about 100 ppm or less, or about 20 ppm or less. These conditions are suitable to achieve the desired stereoselectivity. A 5% ammonium chloride solution (462 kg) was added slowly while maintaining the temperature below 10° C. The aqueous layer was then separated, the pH was adjusted to pH 7-8 by the addition of 6N hydrochloric acid and the mixture was extracted with methyl t-butyl ether (2×145 kg). The combined organic extracts were washed sequentially with 1N sodium hydroxide solution (330 kg) and 20% sodium chloride solution (2×330 kg). THF (767 kg) was then added and the solution was distilled to a residual volume of 165 L. Toluene (567 kg) was added and again the mixture was distilled to a volume of 165 L. The mixture was heated to 85-90° C. until complete dissolution was achieved and then cooled to 20-30° C. to crystallize the product. The solids were collected by filtration, washed with toluene (2×41 kg) and dried at 50-60° C. under vacuum. Yield was 78%. 1H NMR (300 MHz, DMSO-d6) δ 8.3 (d, 1H), 7.4 (d, 1H), 5.2 (s, 1H), 3.5 (m, 1H), 2.4 (m, 4H), 1.4 (s, 3H).
Example 3cis-3-(8-amino-1-bromo-imidazo[1,5-a]pyrazin-3-yl)-1-methylcyclobutanol
Cis-3-(1-bromo-8-chloro-imidazo[1,5-a]pyrazin-3-yl)-1-methylcyclobutanol (27.1 kg), isopropanol (65 kg) and 30% ammonia solution (165 kg) were charged to a suitable vessel. The vessel was sealed and the mixture was heated and stirred for 18 hours at 75 to 85° C. and then cooled. The vessel was vented to a scrubber and water (22 kg) was added. The mixture was concentrated under vacuum to a residual volume of 73-89 L and was then cooled to <5° C. The product was collected by filtration and washed with water (2×108 kg). The product was dried at 40-50° C. under vacuum. Yield was 88%. 1H NMR (300 MHz, DMSO-d6) δ 7.5 (d, 1H), 7.0 (d, 1H), 6.6 (br s, 2H), 5.2 (s, 1H), 3.4 (m, 1H), 2.4 (m, 4H), 1.4 (s, 3H).
Example 4cis-8-amino-3-(3-hydroxy-3-methyl-cyclobutyl)-1-(2-phenyl-quinolin-7-yl)-imidazo[1,5-a]pyrazin-7-ium chloride
This material was prepared by heating OSI-906 with an equivalent of hydrochloric acid in water and then allowing the solution to cool. The solid was filtered from the cooled mixture and dried. The XRPD and DSC suggest a semi-crystalline material. The DSC, XRPD, and 1H NMR (300 MHz, DMSO-d6) of the sample were recorded and are reproduced in FIGS. 1, 2, and 3, respectively.
…………………………………………………………
PATENTS
WO 2010107968
WO 2010129740
WO 2011109572
WO 2011112666
WO 2011163430
WO 2012016095
WO 2012129145
WO 2012149014
WO 2013152252
…………………………………………………………….
The present invention provides for methods of preparing OSI-906 Forms A-G illustrated in Scheme 1 .
…………………………………………………….


THANKS AND REGARD'S
DR ANTHONY MELVIN CRASTO Ph.D
DR ANTHONY MELVIN CRASTO Ph.D
GLENMARK SCIENTIST , NAVIMUMBAI, INDIA
did you feel happy, a head to toe paralysed man's soul in action for you round the clock
need help, email or call me
MOBILE-+91 9323115463
web link
I was paralysed in dec2007, Posts dedicated to my family, my organisation Glenmark, Your readership keeps me going and brings smiles to my family





No comments:
Post a Comment